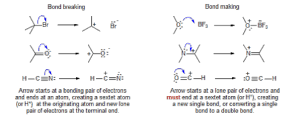A New Branch of Organic Chemistry
A common metaphor used to describe organic chemistry is “the psychology of electrons.” This statement embodies one of the fundamental ideas of organic chemistry: most chemical reactions are driven by the exchange of electrons, rather than protons. Electrons are not in a fixed position like the protons in the nucleus; they follow random orbits around the nucleus, which may cause them to interact with other molecules.
These interactions are most often represented in electron pushing diagrams, such as Figure 1.1. Each line between two atoms represents a shared pair of electrons; likewise, a double line represents two shared pairs of electrons. Electrons normally interact in pairs because electrons have a “quantum spin,” which simplistically means that their movement produces a magnetic pole (much like an electron magnet). However, a lone magnetic pole is highly unbalanced, so two electrons with opposite spins very often pairs to balance each other, much like the magnetic poles of the earth.
A key interest of organic chemists is hydrogenation, or adding hydrogen to a molecule. Adding hydrogen can have the effect of increasing a molecule’s stability, making it a better fuel, or building nanostructures. Dr. Robert Knowles and his lab, however, are studying a type of reaction that will help protons and electrons move in unison, which has the possibility to radically extend the possibilities of reaction chemistry.
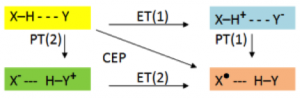
Dr. Knowles studies a type of reaction called Proton-Coupled Electron Transfer (PCET), which is essentially the coupling of a free radical reaction with an acid-base reaction. When a free-radical reaction occurs at the same time as an acid base reaction, the loss of the electron “balances” the loss of a proton. The reasons why this works are still not understood, but scientists have observed that the PCET reaction requires less energy than either electron transfer or proton transfer. In Figure 2 it is easy to see a clear illustration of this idea: to get from the initial (yellow) to final (brown) state, you need to either use a combination of electron transfer and proton transfer, or you can use CEP (another abbreviation for PCET), the most preferred path.
So what’s the use of this? As Dr. Knowles says, “[PCET has] a much, much more expansive energetic range [than other reactions] … You can design catalysts based on these principles that allow you to break the strongest bonds imaginable.” “[Strong bonds]” means that the molecule is highly stable, but also implies that it is lower in energy; higher energy molecules, such as glucose, have less stable bonds than common molecules like CO2. A good analogy might be a spring: strong bonds are like uncompressed springs, while unstable bonds are like compressed springs: the latter are less stable, but can release higher energy if the spring is released/the bond is broken.
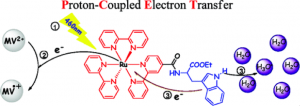
Nature and industry have a strong mutual interest in generating higher energy molecules. Scientists have known for a while that reactions like PCET occur in photosynthesis as well as the splitting of water into oxygen and hydrogen, two high energy processes with high value to the alternative energy business. In Figure 3, a beam of sunlight hits rubisco, a molecule used in photosynthesis, which causes an energy-generating movement of electrons. This is theorized to be a PCET reaction; in the step labeled 3, you can see a proton and electron moving at the same time.
Right now, PCET is not very well understood and somewhat controversial, because there are competing theories for how these reactions work. However, with the work the Knowles lab is doing right now, there is a possibility that organic chemistry could be a very exciting field to study in the next few years.